Correct Answer

verified
2
Correct Answer
verified
Short Answer
How many unpaired electrons are found in each of the following complex ions? -[Co(NH3) 4]2+
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Here are some crystal field representations of d electrons in an octahedral complex:
I) 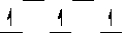 II)
II) 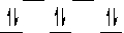 III)
III) 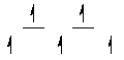 IV)
IV) 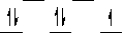 V)
V) 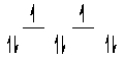 Choose the representation that fits the transition metal atom in the following species:
-K4Fe(CN) 6
Choose the representation that fits the transition metal atom in the following species:
-K4Fe(CN) 6
A) representation I
B) representation II
C) representation III
D) representation IV
E) representation V
G) I) and C)
Correct Answer

verified
Correct Answer
verified
True/False
True or false: Transition metals show great similarities both within a given period and within a given vertical group.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A complex ion is a charged species consisting of a metal ion surrounded by
A) other transition metals
B) hydrogen ions
C) ligands
D) ligands and counter ions
E) none of these
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which model(s) accounts for the magnetism and color of coordination compounds? I.Lewis model II.localized electron model III.crystal field model
A) I
B) II
C) III
D) I,II
E) none accounts for both phenomena
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Fluoride ion ranks low in the spectrochemical series and produces a weak crystal field in complex ions.Based on this information,predict the number of unpaired electrons in CoF64-.
A) 0
B) 1
C) 2
D) 3
E) 4
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The color of a transition metal complex results from:
A) bending vibrations
B) stretching vibrations
C) transition of an electron between d orbitals
D) transition of an electron between an s and a p orbital
E) nuclear magnetic resonance
G) A) and B)
Correct Answer

verified
C
Correct Answer
verified
True/False
Co2+ in water is blue.
B) False
Correct Answer

verified
False
Correct Answer
verified
Multiple Choice
Which of the following complexes would be diamagnetic (all electrons paired) ? Assume the strong-field case for all complexes.
A) [Ni(CN) 6]4-
B) [Ti(CN) 6]3-
C) [Co(CN) 6]3-
D) [Cr(CN) 6]3-
E) none of these
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Name the careful heat treatment of metals that provides the proper combination of strength without too much brittleness.
A) tempering
B) core hardening
C) fire polishing
D) blast furnace
E) direct reduction
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Why do transition metals show a lot of chemical similarities within a given period?
A) The valence s and p electrons affect their chemistry more so than the inner d and f electrons,which do not participate in bonding as easily.
B) The number of electrons within a given period varies only slightly and is sometimes identical because these metals have more than one ionic form.
C) All elements in a given period,including the representative elements,have a lot of chemical similarities due to the gradual increase in atomic number.
D) The transition metals always fill their s and p orbitals first before filling their d orbitals,which affects their chemistry.
E) None of the above is correct.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The coordination theory was proposed by:
A) Bailar
B) Jorgensen
C) Blomstrand
D) Werner
E) none of these
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
When the body adapts to high altitudes,it makes more __________ to adapt to lower oxygen concentrations in the blood.
A) myoglobin
B) iron
C) protein
D) hemoglobin
E) fatty tissue
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many unpaired electrons are there in a complex ion having a d5 electron configuration and an octahedral geometry in the weak field case?
A) 1
B) 2
C) 3
D) 4
E) 5
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Mn(H2O) 43+ (tetrahedral)
A) 0
B) 1
C) 2
D) 4
E) 5
G) A) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Copper(I)complexes would be expected to be colorless.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A metal ion in a high-spin octahedral complex has two more unpaired electrons than the same ion does in a low-spin octahedral complex.The metal ion could be:
A) V2+
B) Cu2+
C) Mn2+
D) Cr3+
E) Co2+
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The transition metal __________ assists insulin in the control of blood sugar and may also be involved in the control of cholesterol.
A) scandium
B) titanium
C) iron
D) nickel
E) chromium
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements concerning the complex ion Co(en) 2Cl2+ is true? (en = ethylenediamine,NH2CH2CH2NH2) ?
A) The complex ion contains Co(I) .
B) The complex ion exhibits cis and trans geometrical isomers,but no optical isomers.
C) The complex ion exhibits two geometrical isomers (cis and trans) and two optical isomers.
D) Since en is a strong field ligand (large ) ,the complex ion is paramagnetic.
E) The geometric isomers of the complex ion have identical chemical properties.
G) A) and D)
Correct Answer

verified
Correct Answer
verified
Showing 1 - 20 of 142
Related Exams