A) an acid
B) a base
C) a salt
D) glucose
E) a neutral substance
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
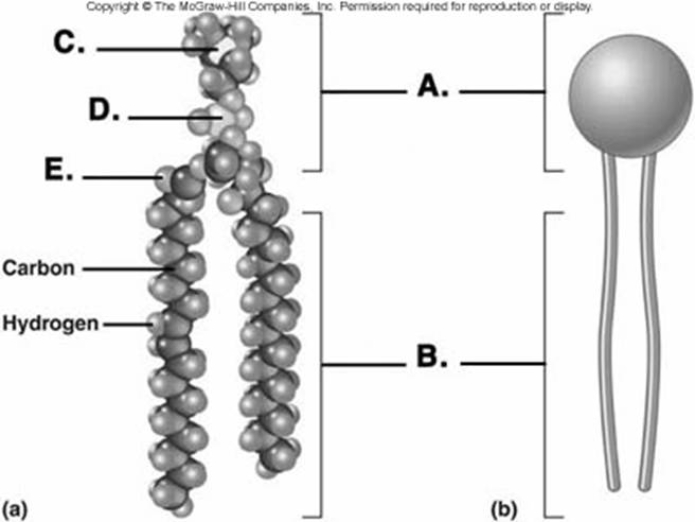 -Phospholipids are important components of the plasma membrane. What does "B" represent on the diagram?
-Phospholipids are important components of the plasma membrane. What does "B" represent on the diagram?
A) phosphorus
B) oxygen
C) nitrogen
D) polar (hydrophilic) region
E) nonpolar (hydrophobic) region
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following chemical reactions best represents the decomposition of ATP?
A) ATP + ADP ATP
B) ADP + ADP + ADP ATP
C) ATP + energy ADP + H2O
D) ADP + Pi + energy ATP + H2O
E) ATP + H2O ADP + Pi + energy
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Carbon dioxide is considered a(n)
A) molecule.
B) compound.
C) molecule and a compound.
D) element.
E) ion.
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
All of the synthesis reactions in the body are called
A) catabolism.
B) hydrolysis.
C) oxidation-reduction.
D) anabolism.
E) dissociation.
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
 -Phospholipids are important components of the plasma membrane. What does "A" represent on the diagram?
-Phospholipids are important components of the plasma membrane. What does "A" represent on the diagram?
A) phosphorus
B) oxygen
C) nitrogen
D) polar (hydrophilic) region
E) nonpolar (hydrophobic) region
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which function of proteins is NOT correctly matched with the example?
A) transport - hemoglobin
B) structure - collagen and keratin
C) regulation - enzymes and hormones
D) protection - packing around organs and glands
E) contraction - actin and myosin in muscles
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is mismatched?
A) ribose--RNA
B) enzyme--protein
C) cholesterol--nucleic acid
D) triglyceride--fat
E) eicosanoid--prostaglandin
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
X-rays can be used to view bones because
A) x-rays pass through bone.
B) x-rays react with bone.
C) x-rays can not pass through bone.
D) bones are less dense than soft tissue.
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Electrons
A) comprise the majority of the mass of an atom.
B) are located in the nucleus of an atom.
C) have a positive charge of one.
D) are the subatomic particles most involved in bonding behavior of atoms.
E) do not participate in the bonding of atoms.
G) All of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The minimum energy required to start a chemical reaction
A) moves in energy surges.
B) results from random molecular movement.
C) comes from ionic energy motion.
D) is elevated by a catalyst.
E) can be lowered by enzymes.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Cobalt-60 is used for radiation treatments of cancer. How many protons does an atom of Co-60 have?
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A cation is
A) a combination of atoms held together by chemical bonds.
B) a positively charged ion.
C) a negatively charged ion.
D) a molecule that conducts electricity when placed in solution.
E) an alteration in the three-dimensional structure of a protein.
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What protein structure results from folding or coiling of a polypeptide chain caused by hydrogen bonds between amino acids?
A) quaternary structure
B) tertiary structure
C) secondary structure
D) primary structure
E) peptide structure
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An atom of chlorine has 17 protons and 18 neutrons. Which of the following statements is true?
A) Chlorine atoms have 18 electrons.
B) Chlorine has a mass number of 35.
C) Chlorine has an atomic number of 18.
D) Chlorine has 35 electrons.
E) Chlorine has an atomic number of 35.
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
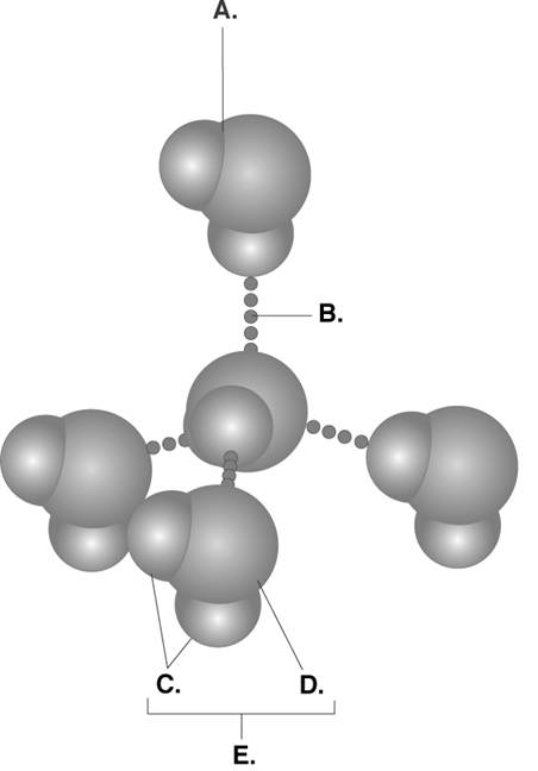 -Water accounts for 50% of the weight of a young adult female and 60% of a young adult male. What kind of bond is found at "B"?
-Water accounts for 50% of the weight of a young adult female and 60% of a young adult male. What kind of bond is found at "B"?
A) hydrogen bond
B) water molecule
C) oxygen atom
D) hydrogen atom
E) polar covalent bond
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the reversible reaction, CO2 + H2O H2CO3 H+ + HCO3- , a decrease in respiration rate will increase the concentration of CO2 in the blood. What will this do to the amount of H+ in the blood?
A) H+ will increase.
B) H+ will decrease.
C) H+ will be unchanged.
E) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is a use of radioactive isotopes?
A) treat cancer
B) diagnose disorders
C) sterilize materials
D) All of these choices are correct.
F) A) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The pH value
A) increases with acidity.
B) is measured on a scale from 0 to 10.
C) is determined by the concentration of hydrogen ions.
D) reflects the sodium content of body fluids.
E) decreases with alkalinity.
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Short Answer
Cobalt-60 is used for radiation treatments of cancer. How many electrons does an atom of Co-60 have?
Correct Answer

verified
Correct Answer
verified
Showing 101 - 120 of 155
Related Exams